Purified water and injection water systems in the biological and pharmaceutical industries
The pharmaceutical water production equipment has its own characteristics, and the equipment accessories are mainly made of sanitary grade stainless steel; The individual device of the equipment can choose to be equipped with pasteurization function; Water supply can choose direct supply circulation mode; Distilled water must be temperature controlled and stored with insulation: automatic control must be comprehensive and have emergency functions for faults, which can maintain the stability and high performance of the equipment for a long time.
Applicable standards:
General requirements for water quality of purified water and injection water in the 2020 edition of the Chinese Pharmacopoeia
American AAMI and ASAIO Hemodialysis Water Standards
GMP standards and specifications, FDA standards, Ep standards
Hemodialysis and Related Treatment Water "Y0572-2005
Common purified water process flow
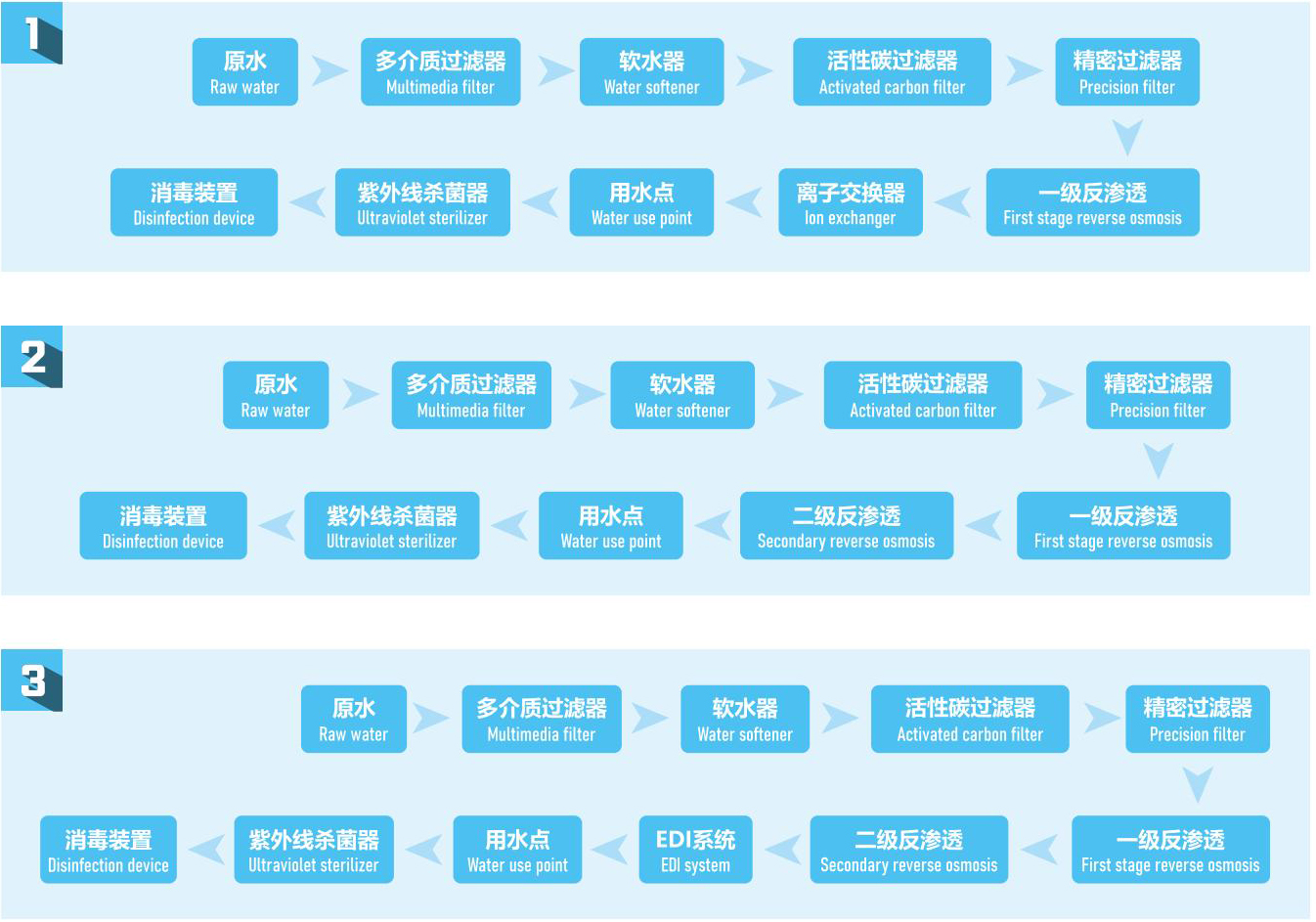
.jpg)








.jpg)
